New Cancer Screening Tests In Asia And Europe
Our latest video report for Volition has gone live and highlights the company’s progress with its colorectal cancer screening triage test in both Asia and Europe.
To see the report, click here
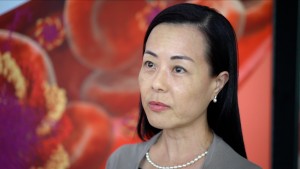
The report features Dr Jasmine Kway, Volition’s Vice President of Asia who says: “We are pleased to be working with Professor Chiu Han-Mo and hope to develop a long-term working relationship. ‘This is an exciting time for Volition as we start our first clinical work in Asia. In addition, we are also taking our first regulatory steps in Asia as we prepare the submission of our CE Marked Nu.QTM Triage product to the Singapore and Taiwan regulatory authorities. Regulatory approval in Singapore and Taiwan would not only make our product saleable in these countries but also potentially in nine other South East Asian markets.”
Volition is a multi-national life sciences company developing simple, easy to use blood-based cancer tests to accurately diagnose a range of cancers. The tests are based on the science of Nucleosomics®, which is the practice of identifying and measuring nucleosomes in the bloodstream or other bodily fluid — an indication that disease is present.
As cancer screening programs become more and more widespread, our products aim to help to diagnose a range of cancers quickly, simply, accurately and cost effectively. Early diagnosis has the potential to not only prolong the life of patients, but also to improve their quality of life.
In Europe, Volition has received Ethical Approval to proceed with the second and final phase of the logistics and pathway design study in Denmark for its Nu.QTM Triage Test. The study, conducted in collaboration with the Hvidovre Hospital and The Danish Research Group on Early Detection of Colorectal Cancer, deals with the practical issues of how the product fits into Denmark’s current national screening program. Results from the study are expected by the end of the third quarter this year and will be presented to the National Screening Committee.
Detection of Colorectal Cancer, deals with the practical issues of how the product fits into Denmark’s current national screening program. Results from the study are expected by the end of the third quarter this year and will be presented to the National Screening Committee.
Volition’s Chief Marketing and Communications Officer, Louise Day commented “We are delighted to receive this timely Ethical Approval for the second phase of the logistics and pathway study. We are hopeful that our collaborators in Denmark will determine the very practical logistics of putting into practice the Nu.QTM Triage Test and provide a smooth, patient-friendly implementation into the Screening Program.”